Measuring length and distance at the molecular level
When we measure length or distance at the molecular level we are talking of a very small measurement.The thickness of a strand of hair while depending on genetics and the weather is approximately from 20 to 180 μm (a μ meter is a millionth of a meter).-------- The diameter of a red blood cell is 6-8 μm...When we talk about molecules microns are too big a unit. We instead use nanometers or Angstroms. 1 Angstrom ( Å ) = 10 nanometers = 10 -10 meters
The graph below shows the first quadrant of a graph where the distance between points on the same axis = 1 Angstrom ( Å ) . The line segments AB and CD are shown in yellow.
-------------->spin on -------->-
spin off
Table of useful conversions for viewing molecules
| 1 Angstrom | 10 -10 meters |
| 1 Nanometer | 10 -9 meters |
| 1 Nanometer | 10 Angstroms |
If you are unfamiliar with Scientific Notation view these tutorials on Edinformatics -- Scientific Notation K-12 Activities
Test your use of Jsmol to measure the length of line segments AB and CD.
You can measure the distance from pt. A to pt B by either counting the number of boxes or by holding your mouse button down over point A --double clicking, then drag the cursor to point B and double click again. The value in Angstroms should show above the line segment.
1: What is the length of line segment AB in Angstroms?
2: What is the length of line segment CD in Angstroms to the nearest hudredth?
About Jsmol
Jsmol is a Javascript framework that uses HTML5 (no java needed) to display interactive 3D molecular structures on Web Browsers. The first time JSMOL is used in each session, it can sometimes be a little slow. Please be patient. Jsmol is currently used by scientists around the world to view molecular structures. The latest version of Mathmol will use Jsmol to view all our molecular structures.
-------------->spin on -------->-
spin off
Measuring the length of a Hydrogen Bond
Above is shown the structure of a Hydrogen Molecule (H2).
Measure the length of the hydrogen bond in Angstroms to the nearest tenth?
MATHMOL
- Activity 1: Measuring length and distance at the molecular level
- Activity 2: Geometry-of-1-Dimension
- Activity 3: Geometry of 2- Dimensions
- Activity 4: Geometry of 3-Dimensions
- Activity 5: Introduction to Molecular Modeling using Jsmol
- Activity 6: The Geometry of Crystals
- Activity 7: Summary Sheet by Students
- Activity 8: What is the Geometry of the Methane Molecule
- Activity 9: Geometry of the Crystal Structure of Ice
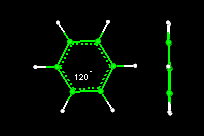
- Activity10:Geometry of the Benzene Molecule